What are the cation and anion that make up the ionic compound CrF4? Give their symbol and charge. | Homework.Study.com

Naming Ionic Compounds. Key Concepts Positively charged ions are called cations Negatively charged ions are called anions The cation is always named. - ppt download
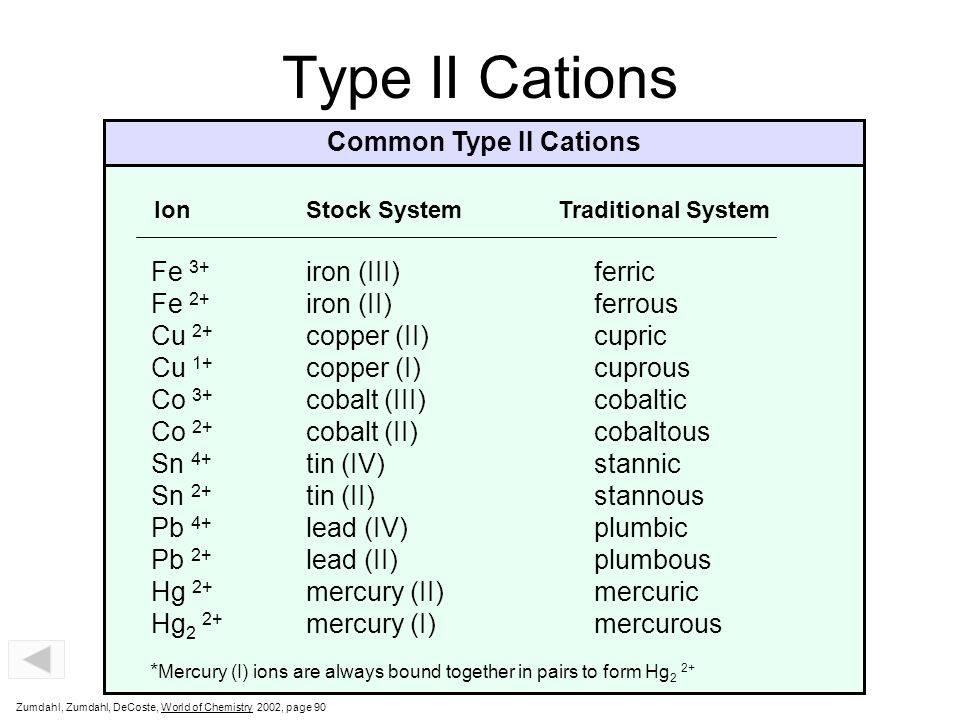
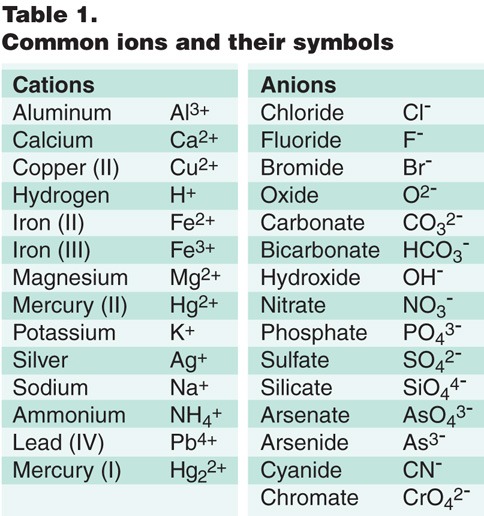
Cations and Anions Common Simple Cations and Anions CationName AnionName* H 1+ hydrogen H 1- hydride Li 1+ lithium F 1- fluoride Na 1+ sodium Cl 1- chloride. - ppt download
Basics, ion exchange, ion exchanger, ion exchange process, softening, demineralization, partial desalination, mixed bed filter, anion exchanger, cation exchanger, exchange resin, Lewatit, regenerating salt, HydroGroup, Hydro-Elektrik GmbH, RWT GmbH
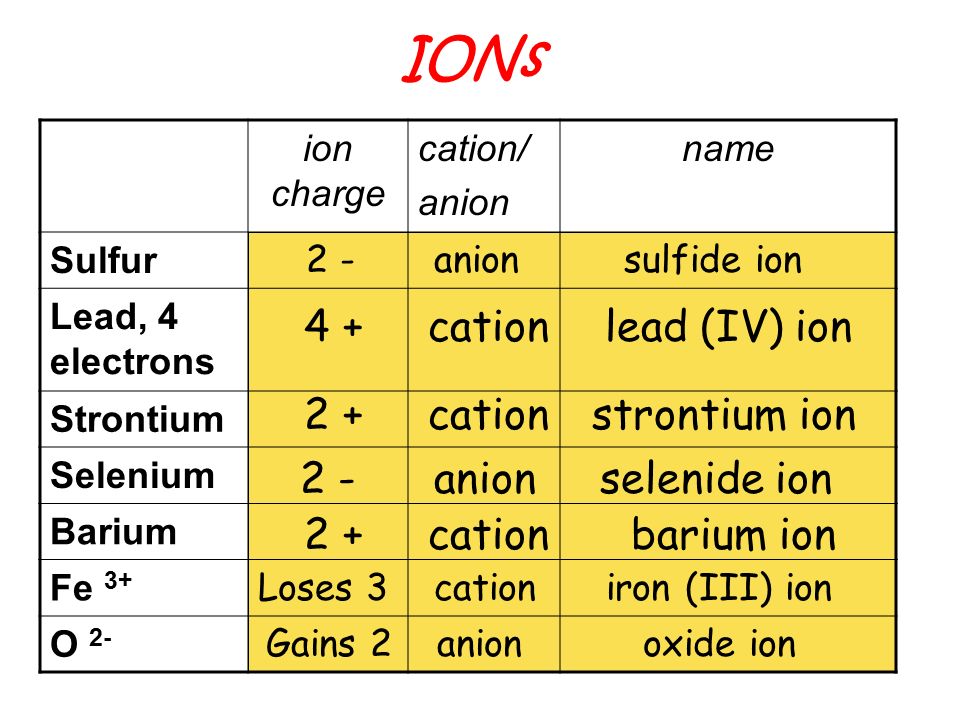
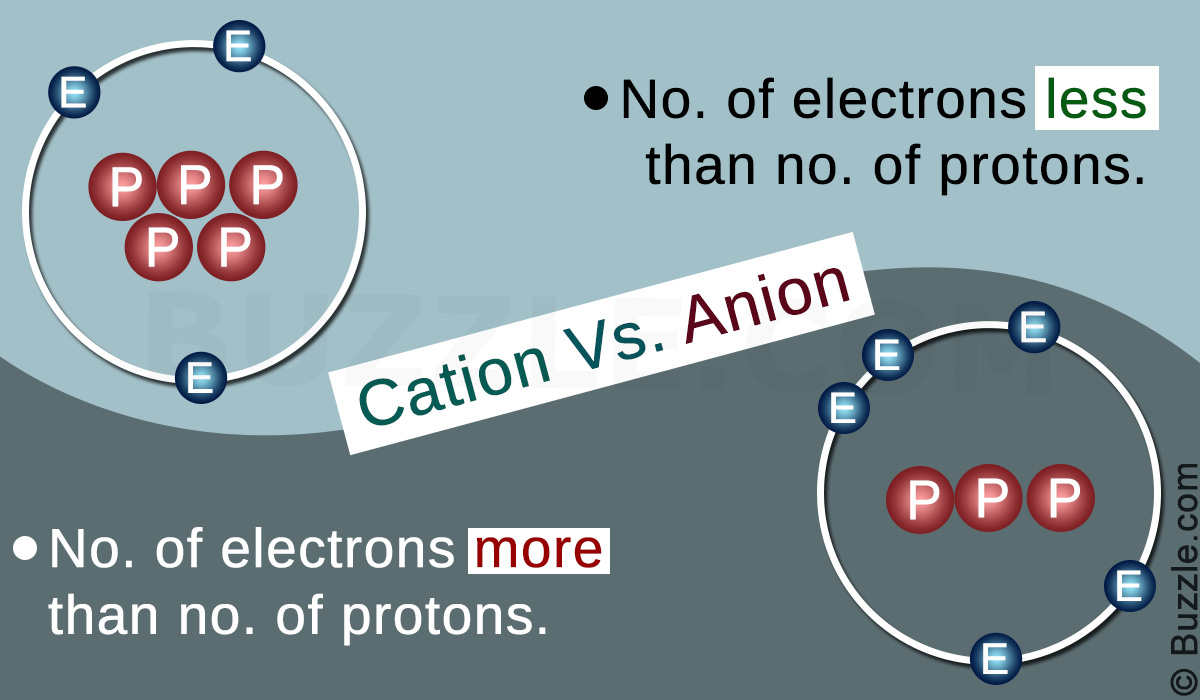
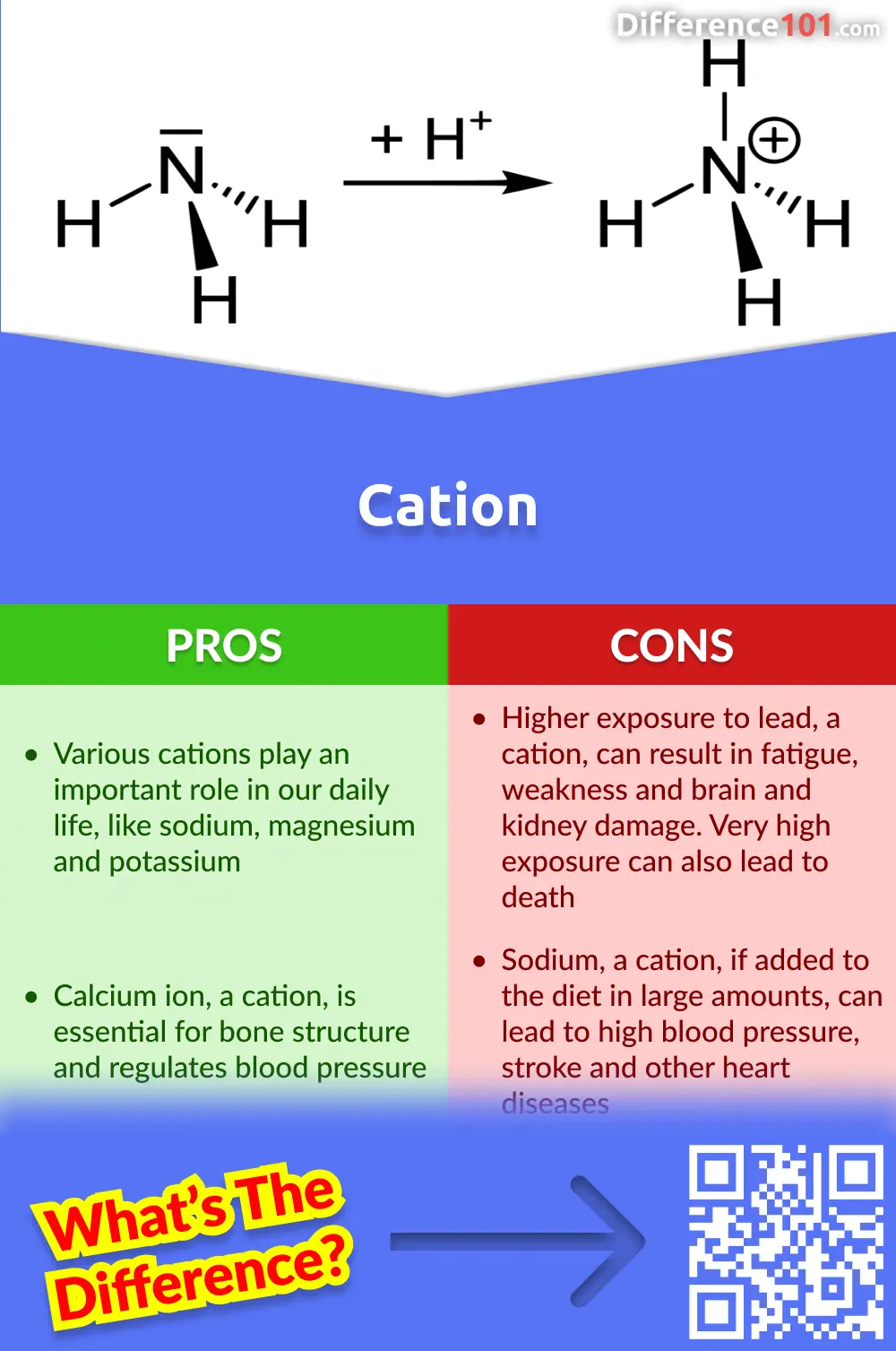
IONS and IONIC COMPOUNDS IONS atoms or groups of atoms that have a positive or negative charge forms when an atom or group of atoms loses or gains electrons. - ppt download

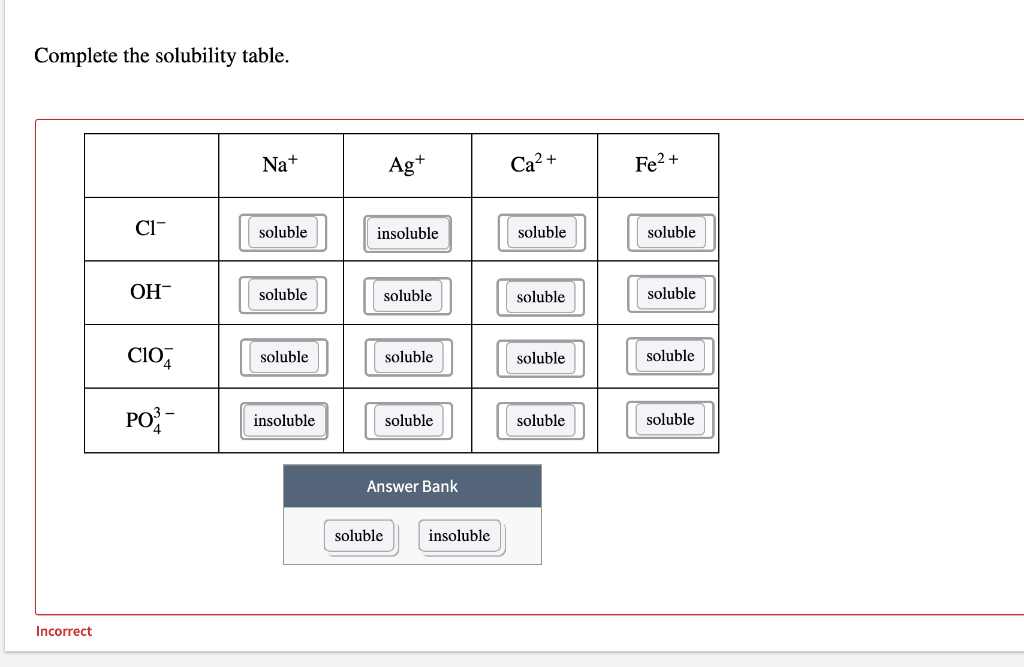
SOLVED:Write the cation and anion in each compound, then determine the correct chemical formula. a. copper (II) sulfide b. nickel (II) nitrate c. iron (II) carbonate d. cobalt (II) sulfate e. iron (
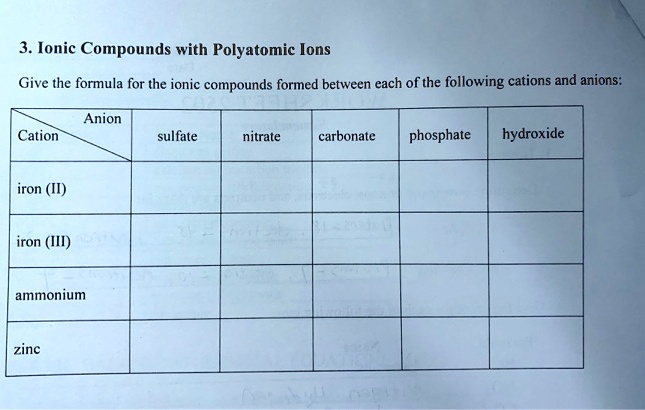
SOLVED: Ionic Compounds with Polyatomic Ions Give the formula for the ionic compounds formed between each of the following cations and anions: Anion Cation sulfate nitrate carbonate phosphate hydroxide iron (II) iron (



:max_bytes(150000):strip_icc()/cation-and-an-anion-differences-606111-v2_preview-5b44daf9c9e77c0037679d52.png)